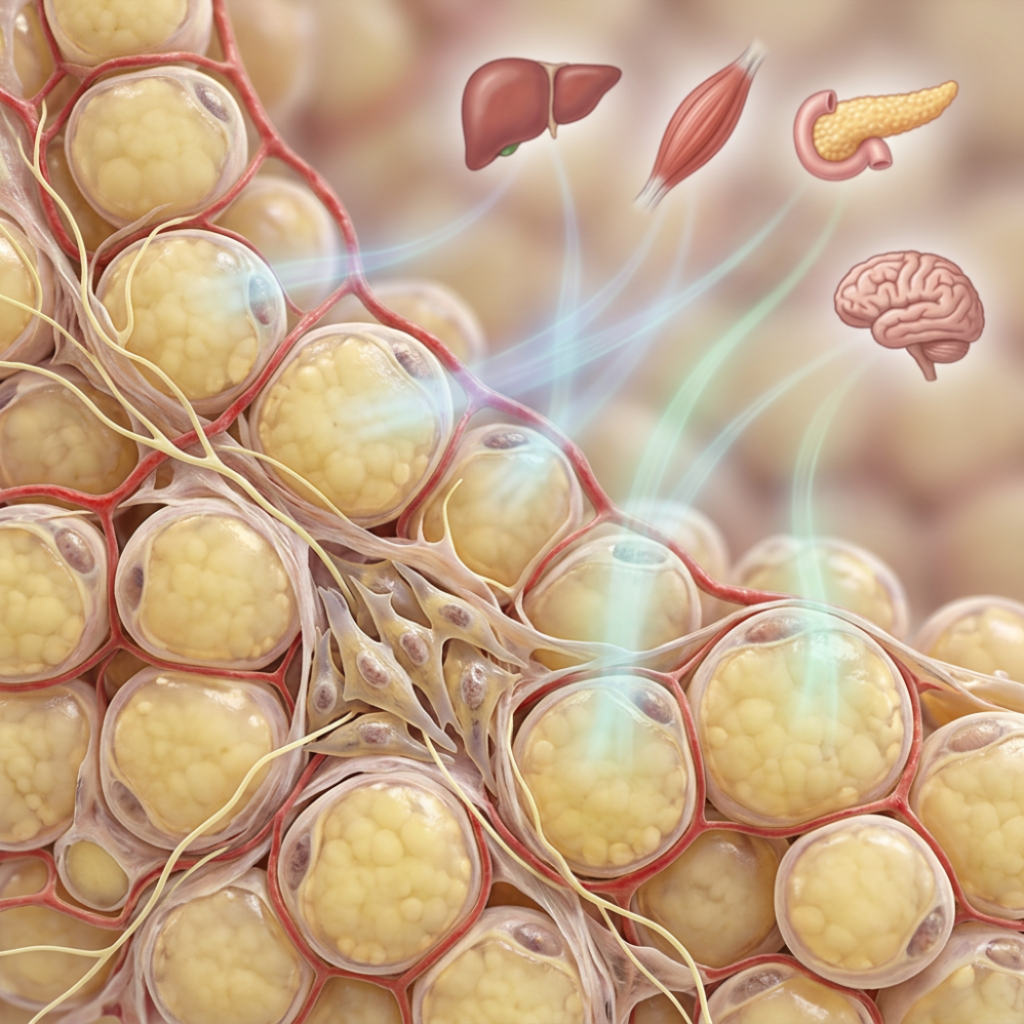
Adipose tissue is more than stored energy. It is an active endocrine organ that releases hormones and signaling molecules into the bloodstream and communicates continuously with the brain, liver, muscle, pancreas, heart, and immune system. When adipose tissue is healthy and well-regulated, this signaling helps keep appetite, insulin sensitivity, inflammation, and vascular tone in balance. When it becomes dysfunctional, as happens in obesity and metabolic stress, the same signaling machinery can drive insulin resistance, chronic inflammation, and a cluster of conditions now described as adiposity-based chronic disease.
TLDR: White adipose tissue secretes hormones and cytokines called adipokines, including leptin and adiponectin, that regulate appetite, insulin sensitivity, cardiovascular risk, and organ function. Brown adipose tissue secretes a different set of signaling molecules called batokines, including FGF21, with beneficial metabolic effects. In obesity and adiposopathy, the endocrine output of fat shifts toward patterns that promote insulin resistance and organ damage. Adipose-derived stem cells and extracellular vesicles interact with these pathways in experimental research, but are not approved treatments for metabolic disease.
Important Disclaimer: Save My Fat does not provide treatments for obesity, diabetes, metabolic syndrome, or cardiovascular disease. No adipose-derived stem cell or exosome product is FDA-approved as a hormone therapy or metabolic treatment. This article summarizes endocrine and regenerative medicine research for educational purposes only and does not constitute medical or legal advice. Patients should work with their healthcare teams to manage weight, blood sugar, blood pressure, and lipids, and to evaluate clinical trial opportunities.
A patient reads that “fat makes hormones” or that “your fat talks to your brain” and wants to understand whether their adipose tissue is helping or hurting them. This is a reasonable question, and the answer is that in healthy states, fat tissue is doing important endocrine work that actively supports metabolic health. The problem is not that fat is an endocrine organ. The problem is what happens when that endocrine function is disrupted by excess, inflammation, and metabolic dysfunction.
Scientists now describe both white and brown adipose tissue as genuine endocrine organs with distinct but complementary roles in whole-body metabolism. White adipose tissue, which makes up the majority of fat in most adults, sends hormonal signals that regulate appetite, insulin action, and inflammation. Brown adipose tissue, which generates heat by burning fuel rather than storing it, sends its own set of signals with broadly beneficial metabolic effects. When either type becomes dysfunctional, the downstream consequences reach far beyond the fat depot itself.
This guide explains the main adipokines and batokines (the signaling molecules from white and brown fat respectively), how adipose tissue communicates with the brain and other organs, what adiposopathy means and how dysfunctional fat drives chronic disease, where adipose-derived extracellular vesicles fit into this endocrine network, and what role adipose-derived stem cells might play in organ-level metabolic signaling in research contexts, with a clear accounting of what remains experimental.
White Adipose Tissue as an Endocrine Organ
From Energy Storage to Hormonal Control
The recognition that white adipose tissue is an endocrine organ emerged gradually through the 1990s and 2000s as researchers identified a growing list of biologically active molecules secreted by adipocytes and adipose stromal cells. A classic review on adipose tissue as an endocrine organ accessible at pubmed.ncbi.nlm.nih.gov/17021375 describes white adipose tissue as a dynamic organ that senses nutrient availability, neural inputs, and hormonal status, then responds by releasing signals that influence energy homeostasis throughout the body. Adipokines, the collective term for these secreted factors, affect appetite, insulin sensitivity, lipid metabolism, blood pressure, immune activity, vascular tone, reproductive function, and bone remodeling.
A broader review of adipokine biology accessible at pubmed.ncbi.nlm.nih.gov/19052866 describes the range of known adipokines and notes that fat tissue cannot be understood simply as a passive repository. Different fat depots, subcutaneous, visceral, epicardial, and perivascular, have different secretory profiles and different metabolic consequences, as documented in a comparative study accessible at academic.oup.com. For background on the cellular biology of adipose-derived stem cells within this endocrine tissue, the patient’s guide to adipose-derived stem cells provides accessible context.
Core Adipokines: Leptin and Adiponectin
Leptin and adiponectin are the most studied and clinically significant adipokines, and their relationship with obesity illustrates the central problem of adipose endocrine dysfunction.
Leptin is produced almost exclusively by adipocytes and is secreted in proportion to fat mass. In lean, healthy individuals, leptin communicates to the hypothalamus (the brain region most directly involved in energy regulation) that long-term energy stores are adequate, reducing appetite and increasing metabolic rate. It acts on pro-opiomelanocortin (POMC) neurons in the arcuate nucleus to suppress food intake and on the pituitary to influence thyroid and reproductive hormones. A detailed account of these brain-adipose interactions is available in a review of adipose-brain interactions accessible at PMC11624209. In obesity, leptin levels are paradoxically elevated because of excess fat mass, but the brain’s response to leptin is blunted. This leptin resistance means that despite abundant circulating leptin, the appetite-suppressing signal is not effectively received, contributing to continued overconsumption.
Adiponectin works in nearly opposite ways from leptin in the context of obesity. In lean individuals, adiponectin circulates at high levels and improves insulin sensitivity by promoting fatty acid oxidation in muscle, reducing hepatic glucose production, and limiting inflammation in vascular walls. Reviews including the Ahima 2006 review accessible at onlinelibrary.wiley.com and the review on adipose as an endocrine organ accessible at sciencedirect.com describe adiponectin as one of the most therapeutically interesting adipokines because of its consistently protective associations. In obesity and insulin resistance, adiponectin levels fall, removing this protective endocrine signal precisely when it is most needed.
Other Adipokines and Their Metabolic Roles
Beyond leptin and adiponectin, white adipose tissue produces a substantial repertoire of additional signaling proteins with distinct metabolic roles. Resistin has been implicated in insulin resistance and inflammation, with higher levels in obese states. Visfatin, also called NAMPT, has roles in metabolism and inflammation. Apelin, omentin, and vaspin have cardioprotective and insulin-sensitizing properties in some experimental contexts. Retinol-binding protein 4 (RBP4) has been linked to insulin resistance and cardiovascular risk. Chemerin influences immune cell recruitment and adipogenesis.
A review of adipokine diversity accessible at pubmed.ncbi.nlm.nih.gov/19052866 and analyses of depot-specific adipokine secretion describe how these signals collectively constitute a complex hormonal language that the brain and peripheral organs are continuously receiving and interpreting. When this language shifts from health-promoting to disease-promoting patterns, the result is what researchers now call adiposity-based chronic disease.
Brown Adipose Tissue and Batokines
Brown Fat as a Distinct Endocrine Organ
Brown adipose tissue (BAT) is metabolically very different from white fat. Its primary function is thermogenesis, generating body heat by uncoupling mitochondrial oxidative phosphorylation from ATP production. In doing so, it burns rather than stores fuel. A 2022 review on brown adipose tissue as an endocrine organ accessible at pubmed.ncbi.nlm.nih.gov/36288452 describes BAT as secreting its own set of biologically active molecules, now called batokines, with effects on other organs that parallel but differ from the effects of white adipose adipokines.
Brown adipose tissue is present in adult humans, particularly in the supraclavicular, cervical, and periaortic depots, and can be activated by cold exposure and certain hormonal and pharmacological signals. In observational studies, higher BAT activity is associated with lower body mass index, better glucose metabolism, and reduced cardiometabolic risk, partly through its direct metabolic effects of burning fuel and partly through its endocrine output.
Key Batokines Including FGF21
Among the batokines identified in research, FGF21 (fibroblast growth factor 21) has attracted the most attention. FGF21 is produced by multiple tissues including the liver, adipose tissue, and muscle, but BAT is a significant source and responder. A 2025 review on emerging insights into brown adipose tissue crosstalk accessible at academic.oup.com describes FGF21 as promoting glucose uptake, lipid oxidation, thermogenesis, and energy expenditure, with effects in white fat, liver, and the central nervous system. Other batokines described in this review and the foundational BAT endocrine review include VEGF-A (which promotes vascularization), BMP8b (involved in thermogenic activation), neuregulin 4 (with hepatoprotective effects), and interleukin-6 (with metabolic effects at physiological levels from exercise-activated muscle and BAT).
The combined endocrine output of brown and white adipose tissue thus shapes systemic metabolism through partially overlapping and partially distinct hormonal signals. In ideal circumstances, high adiponectin and appropriate leptin sensitivity from white fat, combined with active batokine secretion from BAT, create an environment conducive to metabolic health. In obesity and adipose dysfunction, the balance shifts in the opposite direction in both compartments simultaneously.
Brain-Adipose-Organ Communication
The Adipose-Brain Axis
The brain is both the primary target and a major regulator of adipose endocrine function. A review of brain-gut-adipose-tissue communication pathways accessible at PMC3424454 describes the bidirectional hormonal and neural loops through which the brain and adipose tissue monitor each other’s state. Leptin signals long-term energy stores to the hypothalamus. Adiponectin crosses the blood-brain barrier and influences brain-derived neurotrophic factor (BDNF) production, connecting adipose endocrine function to neuroplasticity and potentially to mood and cognition, as described in a review of adipokine effects on brain health accessible at PMC6887733.
Resistin, produced by both adipocytes and immune cells in adipose tissue, can enter the central nervous system and influence the hypothalamic-pituitary-adrenal axis. In obesity, the disruption of adipose-brain communication through leptin resistance, reduced adiponectin, and altered levels of other adipokines may contribute to changes in appetite regulation, mood, cognitive function, and stress response that make sustained weight loss more difficult and that extend the consequences of adipose dysfunction beyond the purely metabolic.
How Adipose Signals Reach the Liver, Muscle, Pancreas, and Bone
The liver receives a particularly direct dose of adipose endocrine output, because visceral fat drains into the portal circulation and its adipokines reach the liver at higher concentrations than the systemic circulation would suggest. Adiponectin reduces hepatic glucose production and fatty acid synthesis. Resistin and inflammatory cytokines from visceral fat promote non-alcoholic fatty liver disease and hepatic insulin resistance. These organ-specific effects help explain why visceral adiposity carries more metabolic risk than subcutaneous adiposity for a given amount of total fat mass.
Skeletal muscle receives adipokine signals from the circulation that influence insulin sensitivity and fatty acid uptake. Adiponectin promotes muscle fatty acid oxidation. Pancreatic beta cells, which produce insulin, are influenced both by circulating adipokines and, as discussed below, by adipose-derived extracellular vesicles. Bone tissue responds to leptin and adiponectin in ways that link energy metabolism with bone density regulation, as reviewed in the adipose review literature.
Adiposopathy and Adiposity-Based Chronic Disease
The concept of adiposopathy, described in clinical and research contexts as pathologically dysfunctional adipose tissue, captures the state in which adipose endocrine output has shifted from health-promoting to disease-driving. A review of adipose tissue inflammation and metabolic dysfunction accessible at PMC6179510 describes how adiposopathy involves elevated pro-inflammatory cytokines, reduced adiponectin, elevated resistin and other harmful adipokines, macrophage infiltration, and fibrosis, all producing a hormonal profile that promotes insulin resistance, endothelial dysfunction, and systemic inflammation.
Adiposity-based chronic disease is a clinical framework that recognizes obesity-related type 2 diabetes, cardiovascular disease, nonalcoholic fatty liver disease, and chronic kidney disease as sharing common pathophysiological roots in adipose dysfunction. This framing is clinically useful because it directs attention to adipose tissue biology rather than treating each downstream disease as a separate problem. The emerging research page on this site tracks where research into adipose biology and its disease connections is most active.
Adipose-Derived Extracellular Vesicles and Organ Crosstalk
How ADEVs Carry Adipose Endocrine Signals
Beyond classical hormone secretion, adipose tissue releases extracellular vesicles, membrane-bound particles carrying proteins, lipids, and RNA molecules that can travel to distant organs and alter gene expression and cell behavior there. Adipocyte-derived EVs (ADEVs) represent a form of endocrine communication that operates in parallel with and overlaps with classical adipokine signaling.
A 2025 review on emerging insights into BAT crosstalk with other organs accessible at academic.oup.com describes how brown fat EVs carry thermogenic proteins and regulatory RNA that can influence gene expression in white fat, liver, and muscle. White adipose-derived EVs carry a different cargo profile that reflects the inflammatory and metabolic state of the source tissue. A review of gut-microbiota-beta-cell interactions accessible at PMC8773909 includes a section on how adipocyte-derived EVs can protect or harm pancreatic beta cells depending on whether the source tissue is healthy or obese.
EVs as Carriers of Adipose Dysfunction
Experimental evidence has shown that EVs from adipose macrophages in obese states carry specific microRNAs, including miR-155, that impair insulin signaling in recipient cells and promote insulin resistance when transferred to liver or muscle. This means that the metabolic consequences of adipose inflammation can spread to other organs through EV-mediated communication before classical inflammatory cytokines reach damaging concentrations. These findings, emerging from animal models and cell culture experiments, suggest that treating adipose EVs as inert particles misses a significant component of how adipose dysfunction propagates systemically.
Lean adipose tissue EVs, by contrast, have been shown in some experimental models to protect beta cells from apoptosis (programmed cell death), to enhance glucose-stimulated insulin secretion, and to promote beneficial gene expression in recipient cells. This protective capacity may be one of the mechanisms through which a healthy adipose endocrine environment supports pancreatic function.
EV Research in Cardiometabolic Disease
Clinical research is beginning to examine adipose-derived EVs as biomarkers of metabolic health and as potential mediators or modulators of cardiometabolic disease. An observational study registered as NCT06408961 (EVOC, EVs in obesity and cardiometabolic disease) is investigating how EV cargo from heart, liver, and fat relates to cardiovascular risk and metabolic health in human populations. This kind of study is at the exploratory phase, characterizing EV biology and its associations with disease, not yet at the therapeutic application phase.
Where ADSCs Fit in This Endocrine Picture
ADSCs and Paracrine Endocrine-Like Signaling
Adipose-derived stem cells are resident in the adipose stromal vascular fraction and are themselves part of the endocrine tissue environment of fat. Their interactions with adipocytes, immune cells, endothelial cells, and nerves influence the local adipose tissue signaling environment. When ADSCs are used therapeutically in experimental settings, their primary mechanism of action appears to be through secreted factors rather than through direct differentiation into new tissue, as reviewed in articles accessible at PMC7787857 and related ADSC reviews.
In the specific context of metabolic organ protection, a study examining ADSC effects on liver injury in a type 1 diabetes model, accessible at PMC8973806, showed that ADSCs attenuated liver damage by reducing mitochondrial stress markers, necroptosis (a form of inflammatory cell death), and macrophage-driven inflammation in the liver. These effects were mediated by paracrine factors rather than direct cell replacement, illustrating how ADSCs can influence organ-level metabolic biology through signaling rather than structural contribution. For the underlying biology of ADSCs within adipose tissue, the patient’s guide to adipose-derived stem cells provides foundational context.
Current Evidence Limits for ADSCs in Metabolic Disease
The preclinical evidence for ADSC effects on metabolic organ function is real and mechanistically coherent but remains far from clinical application. Animal models of diabetes, fatty liver disease, and obesity have produced findings suggesting that ADSC-derived secreted factors can reduce organ inflammation, improve insulin signaling, and protect vulnerable cell populations. Early small human studies in diabetes patients have reported preliminary metabolic signals, as discussed in the metabolic disease article earlier in this series. No ADSC product is approved as a treatment for obesity, type 2 diabetes, or metabolic syndrome.
The appropriate interpretation of this evidence is the same one that applies throughout this series: real scientific findings that justify continued rigorous investigation, not yet sufficient to support clinical adoption outside of registered research protocols. The guide to clinical trials for regenerative medicine explains how to find and evaluate trials for patients interested in research participation.
Banking and the Adipose Endocrine Picture
Banking adipose tissue preserves intact tissue and its cellular and matrix components, including ADSCs with their paracrine signaling capacity and the adipocyte-adjacent populations that participate in adipose endocrine function. The complete guide to adipose tissue banking and the how banking works article explain the mechanics of what is preserved. Banking does not change a person’s current adipokine levels, insulin sensitivity, or cardiometabolic risk profile. It does not replace lifestyle changes, pharmacological management, or surgical interventions that have demonstrated evidence for improving metabolic health.
What banking preserves is the biological material from which future regulated applications might eventually draw. If validated, approved protocols for using ADSCs or adipose-derived EVs in metabolic organ protection emerge, previously banked tissue could become directly relevant. The scientific rationale for such applications is coherent given the biology described in this article. The therapeutic reality is that those applications do not currently exist in approved form.
Frequently Asked Questions
What does it mean that my fat is an endocrine organ, not just storage?
It means that adipose tissue actively secretes hormones and signaling proteins called adipokines that travel through the bloodstream and influence the brain, liver, muscle, pancreas, heart, and immune system. This is not a metaphor; adipocytes produce measurable quantities of leptin, adiponectin, resistin, and many other biologically active molecules whose levels change in response to fat mass, inflammation, and metabolic status. The classic characterization of white adipose as an endocrine organ is described in a review accessible at pubmed.ncbi.nlm.nih.gov/17021375. These adipokine signals shape energy balance and metabolic health in real time throughout adult life.
What do leptin and adiponectin do, and how do their levels change in obesity?
Leptin is produced by fat cells in proportion to fat mass and tells the brain that long-term energy stores are adequate, suppressing appetite and increasing metabolic rate through hypothalamic signaling. Adiponectin is also produced by fat cells but does the opposite in obesity: its levels fall as fat mass increases, removing an insulin-sensitizing, anti-inflammatory signal precisely when it is needed. In obesity, leptin levels are elevated but the brain’s response to leptin is blunted (leptin resistance), so appetite suppression is not achieved despite high circulating leptin. The Ahima 2006 adipose endocrine review at onlinelibrary.wiley.com describes the leptin-adiponectin relationship and its metabolic implications in detail.
What is brown fat, and how can its batokines influence metabolism?
Brown adipose tissue (BAT) is a metabolically distinct type of fat that generates heat by burning fuel rather than storing it. Its distinctive color comes from dense mitochondria. In adults, BAT is present in smaller quantities than white fat but is metabolically significant. BAT secretes its own hormones and signaling molecules called batokines, including FGF21, VEGF-A, neuregulin 4, and others, that positively influence glucose metabolism, fatty acid oxidation, and organ health in experimental systems. The 2022 review on BAT as an endocrine organ at pubmed.ncbi.nlm.nih.gov/36288452 and the 2025 BAT crosstalk review at academic.oup.com describe these batokine effects and their potential metabolic significance.
How does dysfunctional fat, or adiposopathy, contribute to heart, kidney, and metabolic disease?
Adiposopathy refers to pathologically dysfunctional adipose tissue that produces a pro-inflammatory, pro-atherogenic hormonal profile rather than the health-supporting profile of lean, healthy fat. In adiposopathy, adiponectin falls, resistin and inflammatory cytokines rise, macrophages infiltrate the tissue, and the overall adipokine pattern promotes insulin resistance in liver and muscle, endothelial damage in blood vessels, and inflammatory injury in the kidneys. The adiposity-based chronic disease framework groups type 2 diabetes, cardiovascular disease, fatty liver disease, and chronic kidney disease as sharing these common adipose-driven pathophysiological roots.
What are adipose-derived extracellular vesicles and how do they affect beta cells and insulin sensitivity?
Adipose-derived EVs are small membrane-bound particles released by adipocytes and other adipose cells that carry proteins, lipids, and regulatory RNA molecules to distant organs. EVs from lean, healthy adipose tissue have been shown in experimental models to protect pancreatic beta cells and enhance their insulin secretion. EVs from obese, inflamed adipose tissue carry different cargo, including specific microRNAs like miR-155, that can impair insulin signaling in liver and muscle. The BAT crosstalk review and the review on gut-microbiota-beta-cell interactions at PMC8773909 describe these EV-mediated mechanisms in experimental detail. No ADSC-derived or adipose-derived EV product is approved for clinical use in metabolic disease.
Do adipose-derived stem cells fix metabolic syndrome by changing hormones from fat?
No. ADSCs have been shown in preclinical models to attenuate metabolic organ damage through paracrine signaling, particularly in the liver. Small human studies have reported preliminary metabolic signals. But no ADSC product is approved as a treatment for metabolic syndrome, and the mechanism would not be primarily through changing adipokine levels in the classical endocrine sense. ADSC effects in metabolic contexts appear to be primarily through reducing organ inflammation and supporting cell survival, not through reprogramming adipose endocrine output. These are experimental findings that require rigorous clinical validation before any treatment conclusion can be drawn.
If I bank my fat, will it change my endocrine profile or prevent diabetes?
No. Banking preserves adipose tissue in cryogenic storage. It does not interact with your metabolism while frozen, does not change your current adipokine levels, and does not affect your insulin sensitivity, blood glucose, or cardiovascular risk. Your metabolic health today is determined by your current biology, lifestyle, and treatment, not by whether you have banked tissue. Banking is a preservation decision for potential future applications, not a metabolic intervention.
Where can I learn more about adipokines and adipose-organ crosstalk from reputable sources?
The core reviews are accessible through PubMed and PubMed Central. The adipose endocrine organ review at pubmed.ncbi.nlm.nih.gov/17021375 is a foundational reference. The adipokine overview at pubmed.ncbi.nlm.nih.gov/19052866 covers the broader adipokine landscape. The adipose-brain axis reviews are at PMC3424454 and PMC11624209. The BAT endocrine organ review is at pubmed.ncbi.nlm.nih.gov/36288452. The emerging research page on this site tracks current active areas.
Key Takeaways for People Managing Metabolic Risk
Many people think of fat only as accumulated excess calories to be reduced. The biology described in this article reveals something more complex: adipose tissue is an endocrine organ in active hormonal conversation with every major organ system, and the health of that conversation determines much about metabolic risk and protective capacity.
Save My Fat’s priority is to help people understand how adipose tissue communicates with the rest of the body so that choices about lifestyle, medications, trials, and banking are all made with a clear picture of the underlying biology.
The biology-grounded picture:
- White and brown adipose tissue release distinct but complementary sets of hormones and signaling molecules that shape appetite, insulin sensitivity, inflammation, cardiovascular function, and organ health. In adiposopathy, this endocrine output shifts toward patterns that drive insulin resistance and cardiometabolic disease.
- Adipose-derived extracellular vesicles carry the metabolic state of fat tissue to distant organs including the liver, muscle, and pancreas, providing one mechanism by which adipose dysfunction spreads systemically.
- ADSCs and adipose-derived EVs are being studied as tools to modulate metabolic organ injury through paracrine and endocrine-like signaling in experimental models, but they are not approved treatments for obesity, diabetes, or metabolic syndrome.
- Banking adipose tissue preserves a biological resource for future regulated pathways. It does not change current adipokine levels, insulin sensitivity, or metabolic risk today.
The most effective evidence-based approaches for improving adipose endocrine function remain lifestyle modification, pharmacological management of metabolic risk factors, and, where appropriate, consideration of surgical approaches and clinical research participation. Patients interested in research options should discuss those specifically with their endocrinologist or primary care physician. The adipose tissue banking guide, the how banking works article, and the guide to clinical trials for regenerative medicine are available for additional context. Service information including pricing, providers, and family banking is on the site. The about page describes who Save My Fat is.
This article is for educational purposes only and does not constitute medical or legal advice. Legal and medical review including endocrinology expertise is required before publication. Please consult your healthcare providers for all questions about metabolic risk management, obesity treatment, and clinical trial participation.
Last Updated: April 30, 2026







