If you have decided to explore adipose tissue banking, one of the first questions is practical: what does the process actually involve? This guide walks through every step, from the initial consultation to long-term cryogenic storage and potential future retrieval, using published research and clinical trial protocols to explain what happens at each stage.
TLDR: Adipose tissue banking involves a minimally invasive mini-liposuction under local anesthesia (approximately 30 to 60 minutes, outpatient), tissue processing and characterization, cryoprotectant addition, controlled-rate freezing, and long-term storage in liquid nitrogen vapor at approximately -196 degrees Celsius. Published research shows that cryopreserved adipose tissue retains cell viability and stem cell isolation potential after years of storage. The process is regulated under 21 CFR Part 1271 and subject to current good tissue practice (CGTP) requirements. Banking preserves tissue for potential future use in FDA-regulated clinical pathways; it does not guarantee access to any treatment.
Important Disclaimer: Save My Fat does not provide FDA-approved treatments or cures for any disease. Banking adipose tissue does not guarantee eligibility, access, or clinical benefit from any future therapy, clinical trial, or medical program. All content is for educational purposes only and does not constitute medical advice. Patients must consult their own licensed healthcare professionals regarding all medical decisions.
When patients first hear about adipose tissue banking, the concept sounds straightforward: collect fat, freeze it, store it. The reality involves a carefully choreographed sequence of clinical, processing, and quality control steps, each governed by federal regulations and supported by published research.
Overview: The Six Stages of Adipose Tissue Banking
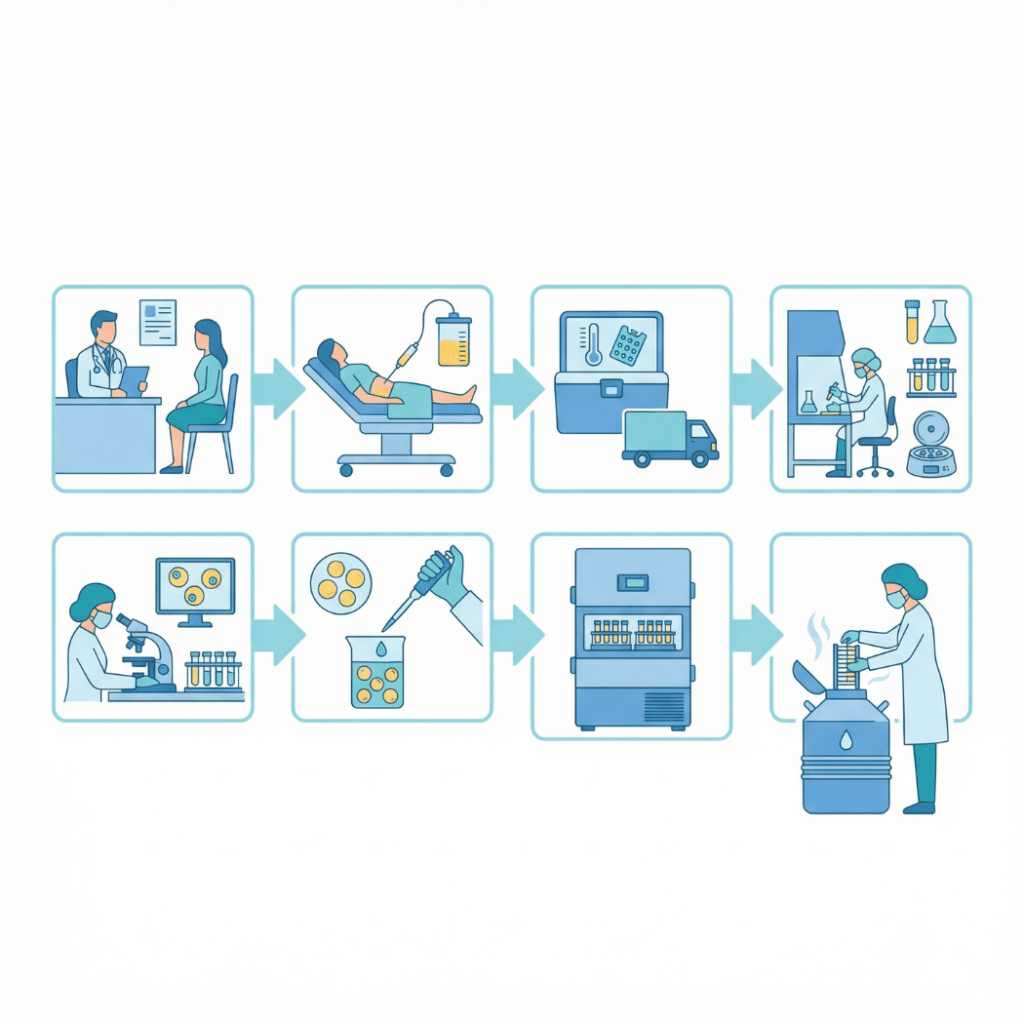
The complete banking process moves through six stages: (1) consultation and eligibility assessment, (2) collection through a mini-liposuction procedure, (3) tissue processing and characterization, (4) cryopreservation including cryoprotectant addition and controlled-rate freezing, (5) long-term cryogenic storage, and (6) retrieval and thawing when needed for an FDA-regulated clinical pathway.
Each stage is described in detail below, with references to published clinical trial protocols and peer-reviewed research. For a broader introduction to why adipose tissue is banked, see the complete guide to adipose tissue banking.
Stage 1: Consultation and Eligibility
The process begins with a consultation to determine whether adipose tissue banking is appropriate for the individual patient. General eligibility includes adults in good general health who can undergo a minimally invasive outpatient procedure under local anesthesia. A physician reviews the patient’s medical history to identify any conditions that might affect the procedure or the quality of the banked tissue.
During the consultation, the physician discusses realistic expectations. Banking is preservation for potential future use in FDA-regulated clinical pathways (clinical trials, expanded access programs, or future approved therapies). It does not guarantee access to any specific treatment. Tissue banking is typically not covered by insurance. For pricing details, visit the pricing page. To find a participating provider, visit the providers page.
Stage 2: The Collection Procedure (Mini-Liposuction)
This is the stage patients ask about most. The collection uses a smaller-scale version of cosmetic liposuction, performed specifically for tissue collection rather than body contouring. It is a minimally invasive outpatient procedure under local anesthesia, meaning the patient is awake throughout. Total procedure time is approximately 30 to 60 minutes (NCT02869061).
Step-by-Step Collection Process
Preparation. The harvest site is cleaned and sterilized. The most common collection sites are the abdomen (highest stem cell yield), flanks, and thighs.
Tumescent anesthesia. A solution called Klein’s solution (saline mixed with lidocaine and epinephrine) is infiltrated into the subcutaneous fat compartment. The lidocaine provides local numbing. The epinephrine constricts blood vessels to minimize bleeding. The solution is infused approximately 15 minutes before aspiration begins, allowing the tissue to become firm (“tumescent”) and anesthetized (NCT02869061).
Incisions. Two small incisions, approximately 5 millimeters (0.5 centimeters) each, are made at the harvest site (NCT02513238 protocol).
Fat aspiration. A 3-millimeter blunt-tipped cannula is inserted through the incisions into the subcutaneous fat layer. The cannula is attached to a syringe and moved through the adipose compartment under gentle suction, mechanically disrupting and collecting the fat tissue. The blunt tip is designed to minimize damage to blood vessels and surrounding structures (NCT03818737 protocol; NCT02513238 protocol).
Volume collected. A typical banking collection harvests approximately 100 to 200 milliliters of lipoaspirate. Clinical trial protocols document that 100 to 2,000 cc can be safely removed in a single liposuction procedure, though banking requires far less volume than cosmetic liposuction (NCT03818737 protocol).
Closure. The small incisions are closed. Sutures are not typically required for 5 mm incisions.
Harvest Site Comparison
| Harvest Site | Estimated SVF Yield per mL | Notes |
| Abdomen | 2-5 x 10^5 SVF cells/mL | Highest yield; most commonly used |
| Flanks | Similar to abdomen | Good alternative |
| Thighs | 1.5-3 x 10^5 SVF cells/mL | Slightly lower yield |
What Does It Feel Like?
Patients are awake under local anesthesia. The tumescent solution numbs the harvest area. Patients may feel pressure, tugging, or movement, but should not feel sharp pain. An oral anti-anxiety medication may be offered pre-procedure at the physician’s discretion.
Recovery After Collection
Most patients experience mild soreness at the harvest site for 3 to 7 days. Bruising is common and typically resolves within 1 to 2 weeks. A compression garment may be recommended for the harvest area. Most patients return to normal daily activities within 1 to 3 days, with no heavy lifting for approximately one week. The incision sites are small (5 mm) and typically heal with minimal scarring.
Documented Risks
Clinical trial protocols document the following risks from the mini-liposuction procedure: bruising (10 to 20%), hematoma (less common), temporary change in skin sensation, swelling, and fluid accumulation. Infection at incision sites is rare. As with any procedure involving local anesthesia, rare allergic reactions are possible (NCT03818737 protocol).
Stage 3: Tissue Processing and Characterization
After collection, the lipoaspirate is transported to the processing facility under controlled conditions. The tissue is washed to remove blood, free lipid (oil), and debris. Sedimentation or gentle centrifugation separates the tissue into three layers: a top oil layer (discarded), a middle intact adipose tissue layer (retained for banking), and a bottom aqueous layer containing blood and tumescent fluid (discarded) (NCT02513238 protocol).
The middle adipose layer is the banking product. This is a critical point: for tissue banking, the adipose tissue is kept intact. It is not enzymatically digested to isolate the stromal vascular fraction (SVF) or adipose-derived stem cells (ADSCs). Keeping the tissue intact preserves the full cellular ecosystem: adipocytes, the complete SVF, and the extracellular matrix. It also maintains the regulatory classification: intact tissue banking may qualify as “minimal manipulation” under FDA regulations, while enzymatic isolation of SVF triggers a different, more restrictive pathway. SVF or ADSC isolation can be performed at a future date if needed for an FDA-regulated clinical application.
For details on the cells within the banked tissue, see What Are Adipose-Derived Stem Cells? and What Is the Stromal Vascular Fraction?. For the full biology of adipose tissue, seeThe Science of Adipose Tissue.
Tissue characterization is performed before freezing, including cell count, viability assessment, and sterility testing. These baseline measurements serve as the reference point for comparing post-thaw quality when the tissue is eventually retrieved.
Important: Banking does not guarantee access to any treatment, clinical trial, or medical program.
Stage 4: Cryopreservation
Cryopreservation is the controlled process of cooling biological tissue to ultra-low temperatures to suspend all biological activity. Without proper cryopreservation, ice crystal formation inside cells would destroy them. For a detailed explanation of the cryobiology, seeHow Cryopreservation Works.
Adding Cryoprotectant
Before freezing, a cryoprotective agent (CPA) is added to protect cells from ice crystal damage. The standard CPA is DMSO (dimethyl sulfoxide) at 10% concentration. DMSO penetrates cell membranes and prevents the formation of large intracellular ice crystals that would rupture cells during freezing. Some protocols add trehalose as a supplemental non-penetrating CPA for added membrane protection (Pu, 2009).
The steps of CPA addition have been well characterized in the cryobiology literature. As described in the published review by Pu (2009), the major steps include adding the cryoprotective agent, cooling to subzero temperatures, storing at cold temperature (usually in liquid nitrogen at -196 degrees Celsius), warming at a later date, and then removing the cryoprotective agent from the cells.
Controlled-Rate Freezing
The tissue is cooled gradually at approximately -1 degree Celsius per minute. This controlled cooling rate allows water to leave cells slowly, preventing large intracellular ice crystals from forming. Cooling too fast causes cells to freeze internally and die. Cooling too slowly causes excessive dehydration and cell death. The rate of -1 degree Celsius per minute is the established standard based on decades of cryobiology research.
A 2025 systematic review confirmed that cryopreservation at -80 degrees Celsius with a cryoprotectant, controlled slow cooling, and fast warming represents the most commonly applied protocol with encouraging outcomes in maintaining tissue survival and histological structure (Bonomi et al., 2025).
Controlled-rate freezers or isopropanol-based passive cooling containers achieve this cooling rate. The tissue passes through the critical temperature range (-15 to -60 degrees Celsius) where ice crystal formation is most likely, and the controlled rate ensures the tissue moves through this zone safely.
Reaching Storage Temperature
Initial cooling typically brings the tissue to -80 degrees Celsius in a mechanical freezer. For long-term storage, tissue is transferred to liquid nitrogen vapor phase at -150 to -196 degrees Celsius. At these temperatures, all biological activity effectively stops.
Stage 5: Long-Term Cryogenic Storage
Tissue is stored in labeled cryovials or cryobags with unique patient identifiers. Storage occurs in liquid nitrogen vapor phase dewars at -150 to -196 degrees Celsius, with continuous temperature monitoring and automated alarm systems. Chain of custody documentation is maintained throughout the storage period. The facility must comply with current good tissue practice (CGTP) requirements under 21 CFR Part 1271.
Storage Temperature Comparison
| Storage Method | Temperature | Duration | Notes |
| Refrigerator | 2-8 degrees C | Hours to days | Not suitable for long-term preservation |
| Mechanical freezer | -80 degrees C | Months to years | Acceptable for medium-term; some cell degradation may occur over years |
| Liquid nitrogen vapor phase | -150 to -196 degrees C | Indefinite (theoretical) | Gold standard for long-term banking; all biological activity halted |
Published Storage Data
Multiple published studies support the viability of long-term cryopreserved adipose tissue. Gonda et al. (2019) reported an average recovery of 67% of the volume frozen from long-term biobanked intact lipoaspirate, with no relationship found between tissue recovery and patient age (PMC, 2019). Devitt et al. (2015) found that patient age did not significantly impact stem cell isolation, viability, or growth after thawing from tissue cryopreserved for up to 1,159 days, concluding that cryopreservation of adipose tissue is an effective long-term banking method for isolation of ADSCs in patients of varying ages (Devitt et al., 2015). A 2025 study of microfragmented adipose tissue stored at -80 degrees Celsius for two years found post-thaw viability of 55.73% compared to 59.75% fresh (no significant difference), with 100% sterility confirmed and histology showing healthy adipocytes with intact membranes (MDPI, 2025).
These results vary by protocol, storage duration, temperature, and cryoprotectant used. Published viability ranges reflect this variation, and results should not be interpreted as guaranteeing specific outcomes for any individual sample.
Stage 6: Retrieval and Thawing
When a patient’s banked tissue is requested for use in an FDA-regulated clinical pathway (such as a clinical trial, expanded access program, or future approved therapy), the retrieval process begins. The facility confirms patient identity and chain of custody. The tissue is thawed using a rapid thawing protocol in a 37 degrees Celsius water bath. Rapid thawing minimizes recrystallization, which is the process of ice crystals reforming during slow warming that can cause additional cell damage.
After thawing, the CPA (DMSO) is removed through serial dilution washing to prevent cytotoxicity, as DMSO can be harmful to cells at body temperature. Published research confirms that double washing efficiently removes DMSO (MDPI, 2025). Post-thaw viability assessment is then performed to characterize the tissue before it is released for the authorized clinical use.
Important caveats: Retrieval occurs only for use in an FDA-regulated clinical pathway. Banking does not guarantee access to any treatment, clinical trial, or medical program. The patient (or their authorized first-degree family member) must provide consent for retrieval. For information on family member eligibility, visit the family page.
Regulatory Framework: How Tissue Banking Is Regulated
Tissue banking in the United States is regulated under 21 CFR Part 1271. Establishments that recover, process, store, label, package, or distribute human cells, tissues, and cellular and tissue-based products (HCT/Ps) must register with the FDA and comply with current good tissue practice requirements (FDA CGTP Guidance).
CGTP requirements govern the methods, facilities, and controls used in the manufacture of HCT/Ps, including all steps in recovery, donor screening, donor testing, processing, storage, labeling, packaging, and distribution. These requirements are designed to prevent the introduction, transmission, or spread of communicable diseases.
A key regulatory distinction applies to adipose tissue banking. The FDA classifies adipose tissue as “structural tissue.” According to the FDA’s 2017 guidance on minimal manipulation and homologous use, “an HCT/P that is cryopreserved and stored in liquid nitrogen vapor, would generally meet the minimal manipulation criterion” (FDA Guidance, 2017; WCG Clinical, 2024). This means that banking intact adipose tissue may be regulated under Section 361 (the less restrictive pathway), provided all four regulatory criteria are met.
By contrast, enzymatic isolation of SVF or culture-expansion of ADSCs is classified as “more than minimal manipulation” and triggers the Section 351 pathway, which requires an Investigational New Drug (IND) application or Biologics License Application (BLA). This distinction is why Save My Fat banks intact tissue rather than processed cell products.
Regulatory Classification Quick Reference
| Product | FDA Classification | Regulatory Pathway |
| Intact cryopreserved adipose tissue | May qualify as minimally manipulated structural tissue | Section 361 HCT/P (if all criteria met) |
| Enzymatically isolated SVF | More than minimally manipulated | Section 351 (requires IND/BLA) |
| Culture-expanded ADSCs | More than minimally manipulated | Section 351 (requires IND/BLA) |
For detailed information about why SVF isolation triggers different regulatory requirements, see What Is the Stromal Vascular Fraction?.
Note: Tissue banking under Section 361 requires registration and CGTP compliance. It does not require pre-market approval. Save My Fat’s service is not “FDA-approved” in the way that a drug or biologic is approved. It is regulated under 21 CFR Part 1271 and subject to CGTP requirements.
Quality Control: What Safeguards Are in Place?
Multiple quality control measures are integrated throughout the banking process. Pre-freeze viability testing establishes a baseline for each sample. Sterility testing (microbiological cultures) confirms the absence of contamination. Cell count and characterization document the cellular content. Continuous temperature monitoring with automated alarms ensures consistent storage conditions. Chain of custody documentation tracks every sample from collection through storage, and unique patient identifiers are applied to all containers to prevent errors.
The facility operates under standard operating procedures (SOPs) for every step of the process, as required by CGTP regulations. When tissue is retrieved for future use, post-thaw viability assessment confirms the quality of the sample before release.
Frequently Asked Questions
How long does the collection procedure take? The mini-liposuction procedure typically takes approximately 30 to 60 minutes. Including preparation and recovery time at the clinic, patients should plan for approximately 2 to 3 hours total.
Is the mini-liposuction painful? The procedure is performed under local anesthesia (tumescent technique). Patients may feel pressure, tugging, or movement, but should not feel sharp pain. Mild to moderate soreness at the harvest site is common for 3 to 7 days after the procedure.
How much fat is collected? A typical banking collection harvests approximately 100 to 200 milliliters of lipoaspirate. This is significantly less than cosmetic liposuction, which may remove several liters.
What happens to the fat after collection? The tissue is washed, separated into layers, and the intact adipose tissue is isolated. It then undergoes characterization (cell count, viability, sterility testing), cryoprotectant addition, controlled-rate freezing, and transfer to long-term cryogenic storage.
How long can my tissue be stored? At liquid nitrogen vapor temperatures (-150 to -196 degrees Celsius), all biological activity effectively ceases. There is no established maximum storage duration at these temperatures. Published studies have documented successful stem cell isolation from tissue cryopreserved for over three years (Devitt et al., 2015).
Will my stem cells survive freezing? Published research shows that ADSCs can be successfully isolated from cryopreserved adipose tissue, with viability rates that vary by protocol, storage duration, and temperature. No protocol guarantees 100% cell survival, but published data consistently shows that viable stem cells can be recovered after long-term storage.
How do I know my tissue is being stored properly? Storage facilities use continuous temperature monitoring with automated alarm systems, chain of custody documentation, unique patient identifiers, and standard operating procedures governed by CGTP requirements under 21 CFR Part 1271.
What if I never need my banked tissue? The tissue remains in storage for as long as the patient maintains their banking agreement. There is no obligation to use banked tissue.
Can my family members use my banked tissue? First-degree family members may potentially qualify to use banked cells under certain guidelines. This requires additional screening and would follow applicable FDA regulatory pathways. Visit the family page for details.
Is tissue banking FDA-regulated? Yes. Tissue banking in the United States is regulated under 21 CFR Part 1271. Facilities must register with the FDA and comply with current good tissue practice (CGTP) requirements.
What is the difference between banking intact tissue and banking isolated stem cells? Banking intact tissue preserves the complete cellular ecosystem (adipocytes, SVF, ECM) without enzymatic processing. This approach may qualify as “minimal manipulation” under FDA regulations. Isolating SVF or culturing ADSCs involves additional processing that triggers more restrictive regulatory requirements under Section 351.
How much does adipose tissue banking cost? For current pricing information, visit the pricing page. Tissue banking is typically not covered by health insurance.
Key Takeaways
The Process Is Defined and Regulated
- Adipose tissue banking follows a six-stage process governed by federal regulations and informed by published clinical research
Collection Is Minimally Invasive
- The mini-liposuction is an outpatient procedure under local anesthesia, taking approximately 30 to 60 minutes with a recovery period of days, not weeks
Intact Tissue Banking Has Regulatory Significance
- Banking intact adipose tissue (without enzymatic digestion) may qualify as minimal manipulation under FDA guidelines, which is why this approach is used rather than isolating SVF or ADSCs
Published Research Supports Long-Term Viability
- Multiple studies demonstrate that cryopreserved adipose tissue retains cell viability and stem cell isolation potential after years of storage
Quality Control Is Built Into Every Stage
- Viability testing, sterility testing, continuous monitoring, and chain of custody documentation ensure accountability from collection through storage
Banking Is Preservation, Not Treatment
- Tissue banking preserves adipose tissue for potential future use in FDA-regulated clinical pathways; it does not guarantee access to any treatment, clinical trial, or medical program
Learn More About Adipose Tissue Banking
Before contacting Save My Fat: Adipose tissue banking is a preservation service for potential future opportunities, not a treatment or cure for any disease. Outcomes cannot be guaranteed. All medical decisions must be made with a licensed healthcare provider.
Visit the how stem cell banking works page for an overview.
For the complete guide, see What Is Adipose Tissue Banking?.
For current pricing, visit the pricing page.
Healthcare providers can learn more on the providers page.
Learn about Save My Fat at the about page.
Last updated: March 12, 2026