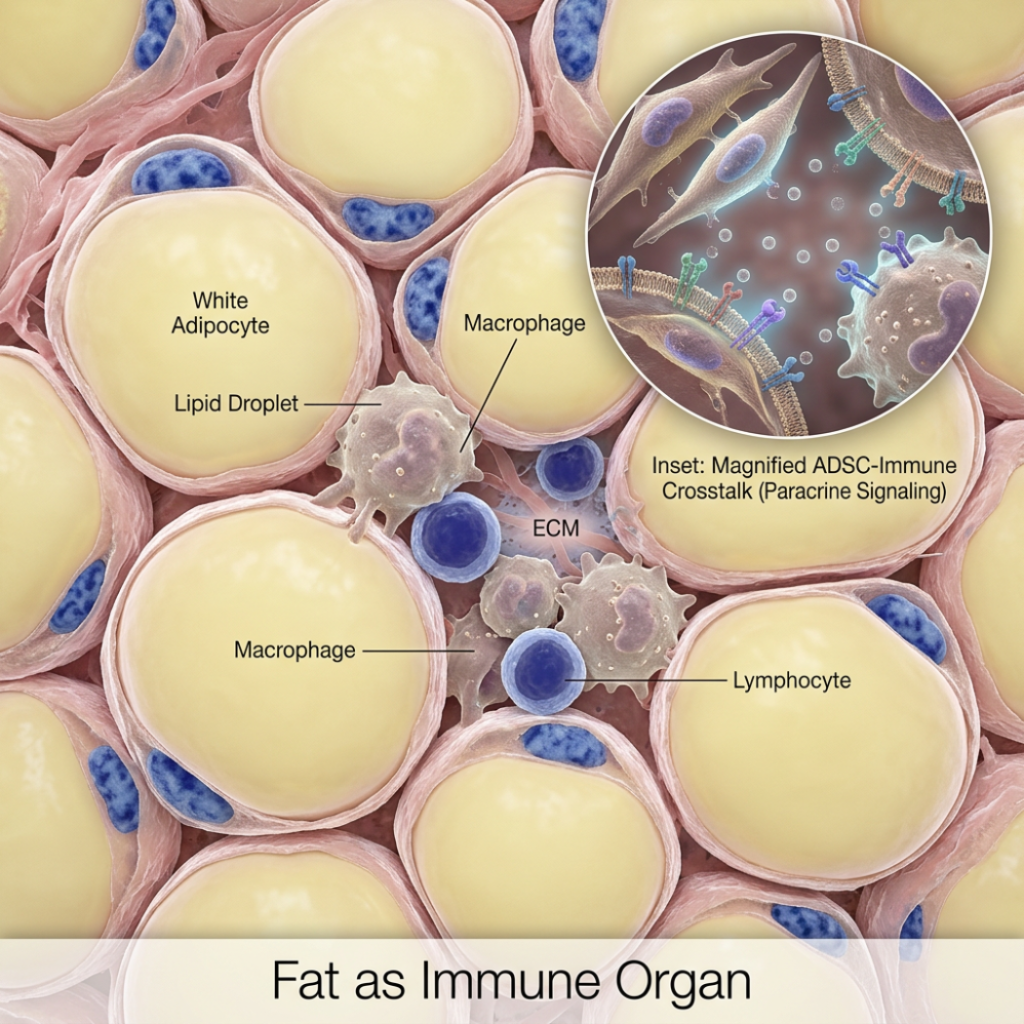
Fat is not just a storage depot. Adipose tissue contains immune cells, produces hormones and signaling molecules called adipokines, and is in constant conversation with the immune system across the entire body. When adipose tissue is healthy, this active role helps maintain metabolic balance and immune homeostasis. When it becomes inflamed, as happens in obesity and metabolic disease, it can drive a chronic low-grade inflammatory state that interferes with insulin signaling and contributes to a range of systemic conditions. Understanding this changes what it means to think about your fat.
TLDR: Adipose tissue is a surprisingly active immune organ. It houses innate immune cells like macrophages and innate lymphoid cells as well as adaptive immune cells like T cells, B cells, and regulatory T cells, and it releases adipokines and cytokines that shape whole-body metabolism and immunity. In obesity and metabolic disease, adipose immune balance shifts toward chronic low-grade inflammation that drives insulin resistance. Adipose-derived stem cells can modulate immune responses in experimental settings through paracrine and contact-dependent mechanisms, but that does not make them a general immune cure.
Important Disclaimer: Save My Fat does not provide immune therapies, autoimmune disease treatments, or off-protocol ADSC infusions. No adipose-derived stem cell product is FDA-approved to treat autoimmune disease, metabolic syndrome, or chronic inflammation as a primary indication. This article summarizes immunology and regenerative medicine research for educational purposes only and does not constitute medical or legal advice. Patients should work with their healthcare teams to manage immune and metabolic conditions and to evaluate any clinical trial or investigational therapy.
Many people hear that fat “makes inflammation” or that “stem cells calm the immune system” and feel simultaneously interested and confused. These are not empty marketing phrases. They trace back to real biology, but the biology is substantially more nuanced than clinic advertising typically communicates. Adipose tissue has a dual immunological role: it supports metabolic and immune health in lean, fit conditions, and it becomes a driver of metabolic dysfunction when it is chronically inflamed. Adipose-derived stem cells have measurable immunomodulatory properties in laboratory and animal research. Neither of these things means that fat is simply a problem to be fixed or that stem cells solve immune dysregulation.
Scientists have spent the last two decades redesigning how they think about adipose tissue. What was once understood primarily as an energy reservoir is now recognized as an endocrine organ producing hormones that regulate appetite and insulin sensitivity, and as an immune organ housing a resident population of innate and adaptive immune cells that shape systemic metabolism. That recharacterization has direct relevance to anyone thinking about conditions tied to inflammation, from metabolic syndrome to autoimmune disease to the biology of aging.
This guide covers what immune cells live in adipose tissue in healthy and obese states, how adipose inflammation drives insulin resistance, how adipose-derived stem cells interact with immune pathways in experimental models, what that means and does not mean for patients, and how tissue banking fits into a realistic long-term view of these questions.
Adipose Tissue as an Immune Organ
From Storage Depot to Immune Organ
The shift in scientific understanding of fat began with studies showing that adipose tissue contained large numbers of immune cells even in lean, healthy individuals. A foundational review accessible at PMC4340740 describes adipose tissue as an active immunological organ in which resident immune cells communicate bidirectionally with adipocytes and with the systemic immune system through cytokines, adipokines, and direct cell contact. In lean adipose tissue, this communication supports insulin sensitivity, tissue homeostasis, and anti-inflammatory balance. In obese adipose tissue, the communication patterns shift substantially toward chronic inflammation.
The review at PMC5199693 on innate and adaptive immunity in obesity-associated metabolic disease describes how the immune changes in adipose tissue are not secondary effects of metabolic dysfunction but active contributors to it. The inflammatory cells that accumulate in obese fat are capable of producing cytokines that directly impair insulin signaling in adipocytes, hepatocytes, and skeletal muscle, linking adipose inflammation mechanistically to systemic insulin resistance.
Innate Immune Cells in Adipose Tissue
The innate immune system provides rapid, non-specific responses to injury and infection. In lean, healthy adipose tissue, resident innate immune cells are dominated by cells with anti-inflammatory or homeostatic properties. Adipose tissue macrophages in the lean state are often characterized as M2-like (a functional descriptor indicating a more anti-inflammatory, tissue-repair-oriented activation state), and they coexist with eosinophils, innate lymphoid cells (ILCs), and mast cells that help maintain the tissue environment favorable to insulin sensitivity.
A comprehensive review of innate immune cells in adipose tissue accessible at PMC8787575 describes how this balance shifts in obesity. As fat depots expand and adipocytes grow larger (hypertrophy), they can become hypoxic and begin to die. Dying adipocytes trigger the infiltration of pro-inflammatory M1-like macrophages, which cluster around dead adipocytes to form structures called crown-like structures (CLS) visible in tissue sections from obese individuals. These M1-like macrophages produce high levels of tumor necrosis factor-alpha (TNF-alpha), interleukin-6 (IL-6), and interleukin-1 beta (IL-1 beta), cytokines that interfere with insulin receptor signaling and promote further inflammatory recruitment. Neutrophils and mast cells also increase in obese adipose tissue, adding additional pro-inflammatory mediators.
A 2021 Frontiers in Immunology article on adipocytes and innate immunity, accessible at frontiersin.org, adds another dimension: adipocytes themselves express pattern-recognition receptors (molecular sensors for bacterial and viral components) and MHC molecules (cell surface proteins that present antigens to immune cells). This means adipocytes are not just bystanders to the immune conversation in fat tissue; they are active participants.
Adaptive Immune Cells in Adipose Tissue
The adaptive immune system, comprising T cells and B cells, mounts specific, long-lasting responses and is increasingly recognized as a major contributor to metabolic regulation in adipose tissue. A review on the adaptive immune system as a regulator of adipose tissue and metabolism, accessible at Wiley, describes the immunological complexity of fat as extending well beyond the macrophage-focused view that dominated earlier research.
In lean adipose tissue, regulatory T cells (Tregs, a suppressive T cell subset that limits immune overreaction) and type 2 helper T cells (Th2) are relatively abundant and contribute to anti-inflammatory conditions that support insulin sensitivity. In obesity, the balance shifts: Th1 cells (which drive inflammatory responses) and CD8+ cytotoxic T cells increase, Th17 cells (which produce the pro-inflammatory cytokine IL-17) become more prevalent, and Tregs decrease. A 2023 review of adipose tissue lymphocytes in obesity accessible at oaepublish.com details how these T cell shifts precede and contribute to macrophage infiltration in the chronology of obesity-related adipose inflammation.
B cells, which produce antibodies and provide help to T cells, also reside in adipose tissue. In obesity, B cell populations and autoantibody production in adipose tissue increase in animal models, and antibodies against fat cell components may contribute to local inflammation. The coordination between adipose macrophages and T cells is described in a 2024 review accessible at PubMed, which characterizes the adipose macrophage-T cell network as a central organizing feature of metabolic inflammation. For foundational context on what adipose-derived stem cells are within this tissue environment, the patient’s guide to adipose-derived stem cells explains how ADSCs exist alongside these immune populations.
How Adipose Inflammation Links to Metabolic Disease
Chronic Low-Grade Inflammation and Insulin Resistance
The term “chronic low-grade inflammation” describes an immune state distinct from the acute inflammation of an injury or infection. In obesity-related adipose inflammation, there is no single trigger like a pathogen; instead, there is a sustained, lower-intensity inflammatory signal arising from the physical and metabolic stress on expanding fat tissue. The review at PMC8787575 describes how multiple factors contribute to triggering this state: adipocyte hypertrophy creating mechanical stress and hypoxia, endoplasmic reticulum stress within overloaded adipocytes, and molecules from the gut microbiome crossing into circulation during metabolic dysfunction.
The inflammatory cytokines produced by this process, particularly TNF-alpha and IL-6 from infiltrating macrophages, interfere with insulin signaling by activating inflammatory kinases that phosphorylate insulin receptor substrate proteins at inhibitory sites, essentially inserting an inflammatory brake into the insulin response pathway. This mechanism connects what happens in adipose tissue directly to insulin resistance in peripheral tissues. Improving adipose immune balance, through weight loss, physical activity, or potentially through therapeutic interventions, can reduce these inflammatory signals and improve insulin sensitivity.
Immune Cell Networks and Metabolic Regulation
The adipose macrophage-T cell interaction is not just a bystander phenomenon. The 2024 review at PubMed and a Nature Reviews analysis on resident and migratory adipose immune cells controlling systemic metabolism describe adipose immune cells as active regulators of whole-body energy homeostasis in health. In lean adipose tissue, resident M2-like macrophages facilitate lipid turnover, support thermogenesis in brown adipose tissue, and clear cellular debris that would otherwise trigger inflammation. In obesity, the shift toward inflammatory macrophage and T cell dominance disrupts not just insulin sensitivity but also thermogenic capacity, contributing to a metabolic environment that tends to perpetuate excess fat storage.
Adipocytes as Immune-Like Cells
A dimension of adipose immunology that often surprises people is that adipocytes themselves behave like immune cells in important ways. Beyond responding to inflammatory signals, adipocytes produce leptin, a hormone that among its many functions acts on immune cells to promote inflammatory responses. They produce adiponectin, which has anti-inflammatory and insulin-sensitizing properties and declines with obesity. In obese states, the shift toward high leptin and low adiponectin changes the immune environment in ways that favor inflammation and reduce immune tolerance. Adipocytes also produce complement proteins and express toll-like receptors (TLRs), pattern-recognition receptors that detect bacterial components and trigger innate immune activation.
How Adipose-Derived Stem Cells Talk to the Immune System
Core Immunomodulatory Mechanisms
Published reviews of ADSC biology, including a comprehensive analysis of adipose-derived MSC impacts at PubMed and a 2021 Scientific Reports study on immune modulation via adipose-derived MSCs at Nature, describe a well-characterized set of immunomodulatory mechanisms that operate through paracrine signaling and direct cell contact.
At the cytokine level, ADSCs in inflammatory environments secrete increased amounts of anti-inflammatory molecules including interleukin-10 (IL-10), transforming growth factor beta (TGF-beta), and interleukin-1 receptor antagonist (IL-1Ra), which can dampen the pro-inflammatory cytokine cascade. They also produce prostaglandin E2 (PGE2) and indoleamine 2,3-dioxygenase (IDO), two mediators that suppress T cell proliferation and promote the development of Tregs. At the cell surface, ADSCs express programmed death-ligand 1 (PD-L1), which engages the PD-1 checkpoint on T cells to reduce their activation.
The overall picture is of cells that are immunologically alert: they respond to an inflammatory environment by increasing their immunosuppressive output. This has been described as immune “licensing,” where exposure to pro-inflammatory signals such as IFN-gamma and TNF-alpha makes ADSCs more potently immunomodulatory, not less. This is conceptually important for understanding why ADSC effects in highly inflammatory disease contexts may differ from effects in lower-inflammation conditions.
Effects on Specific Immune Cell Populations
In terms of specific immune populations, ADSC research has documented the following patterns in experimental models. For T cells, ADSCs suppress the proliferation of activated helper and cytotoxic T cells, reduce Th1 and Th17 cytokine production, and increase the generation of Tregs, the suppressive T cell subset that is reduced in obesity and in many autoimmune conditions. For B cells, ADSCs can reduce B cell proliferation and immunoglobulin production in some experimental systems. For macrophages, ADSCs and their extracellular vesicles can promote a shift from M1-like (pro-inflammatory) to M2-like (tissue-repair-oriented) activation states, reducing TNF-alpha and increasing IL-10 production. For dendritic cells, which are the primary cells that activate the adaptive immune system, ADSCs can interfere with maturation and reduce their ability to stimulate T cell responses.
A study specifically examining sex-dependent differences in ADSC immunomodulation, accessible at Nature, illustrates an important practical point: donor characteristics influence immunomodulatory potency. ADSCs from female and male donors showed differences in their immunosuppressive capacity in that study, pointing to the broader principle that ADSC biology is not uniform across donors. Cell preparation method, donor age, donor metabolic status, and the inflammatory environment in which the cells are used all contribute to variability in immunomodulatory output. The emerging research page on this site tracks where ADSC immunomodulation research is currently active.
Context Determines Effect
One of the most important concepts for understanding ADSC immunomodulation is context dependence. These are not cells that uniformly suppress the immune system. They are cells that respond to their environment and can shift their secretory profile based on the signals they receive. In a highly inflammatory environment, they become more immunosuppressive. In a lower-inflammation environment, their effects may be less pronounced. This context dependence is scientifically coherent, but it also means that extrapolating from experimental models to specific patient situations requires care.
It also raises the question of dose, route, and timing. The immune effects observed in in vitro experiments involve defined cell-to-cell ratios in controlled environments. What happens when a heterogeneous cell preparation is introduced into a complex systemic disease context, with its own pre-existing immune architecture, is substantially harder to predict. This is precisely why early human ADSC trials in immune-mediated conditions focus first on safety and tolerability before attempting to draw conclusions about immune mechanism.
What This Does and Does Not Mean for Patients
Evidence from Preclinical and Early Clinical Work
The preclinical evidence for ADSC immunomodulation is robust in experimental systems. Animal models of autoimmune conditions including lupus, rheumatoid arthritis, inflammatory bowel disease, and type 1 diabetes have shown that ADSC administration can reduce tissue inflammation, lower autoantibody titers, improve organ function, and extend survival in some models. These findings provide the scientific rationale for moving into human trials.
Human trials in immune-mediated conditions, including graft-versus-host disease, Crohn’s disease, and the inflammaging trial NCT05827757 described in an earlier article in this series, are at early phases focused primarily on safety and exploratory immune marker changes. The consistent finding across these early trials is that ADSC administration appears generally tolerable and that some inflammatory markers change in directions consistent with the experimental predictions. What has not yet been established in well-powered, controlled human trials is whether these immune changes translate into meaningful, durable clinical benefit for specific conditions. For context on how clinical trials are structured and what early-phase results mean, the guide to clinical trials for regenerative medicine explains the framework.
Why “Anti-Inflammatory” Is Not a Magic Word
The term “anti-inflammatory” is used so broadly in wellness marketing that it has largely lost its specificity as a scientific descriptor. When applied to ADSCs, it carries a real meaning backed by experimental evidence. But that meaning is conditional in ways that marketing rarely acknowledges.
Immune suppression, even when it reduces inflammation, carries its own risk profile. Reducing immune activity that is necessary for pathogen defense creates vulnerability to infection. Altering the immune environment around developing tumors could theoretically influence tumor behavior in complex ways. The comprehensive ADSC impact review at PubMed acknowledges these dual-edged concerns explicitly. In legitimate clinical trials, these risks are monitored, adverse events are reported, and the risk-benefit balance is assessed within a structured framework. In commercial clinical settings outside of trials, this monitoring infrastructure is largely absent.
Banking and Future Immune Pathways
Banking adipose tissue preserves cells with immunomodulatory potential in their native tissue context. The complete guide to adipose tissue banking and the how banking works article explain what the banking process involves. Banking does not provide a ready-to-use immune therapy product. It does not change a person’s current immune status. Any future use of banked tissue for immune-modulating purposes would require its own regulatory and clinical development pathway.
What banking does is preserve a biological resource at its current state for potential future opportunities in pathways that do not yet exist in finalized form. Patients with autoimmune conditions, inflammatory diseases, or metabolic concerns who are considering banking should understand this clearly and should not allow the prospect of future immune applications to substitute for evidence-based management of their conditions today.
Practical Questions for Patients About Immunity, Fat, and ADSCs
Questions About Adipose Inflammation and Health
Patients who are concerned about chronic inflammation and adipose tissue health should be asking their clinicians how their weight, waist circumference, and metabolic lab values relate to markers of adipose inflammation such as fasting insulin, C-reactive protein, and adiponectin levels. They should ask whether lifestyle interventions, including dietary changes, increased physical activity, and improved sleep quality, have been shown to reduce adipose inflammatory markers and improve insulin sensitivity, and what the evidence shows for those interventions in their specific situation. Standard medical treatments for metabolic conditions, from SGLT2 inhibitors to GLP-1 receptor agonists, now have substantial evidence for anti-inflammatory effects in adipose tissue and systemically, and those treatments deserve consideration alongside any investigational approach.
Questions About ADSC-Based Immune Modulation
For patients who are evaluating ADSC-based interventions for immune conditions, the critical questions are whether the specific intervention has been tested in randomized, controlled trials for the specific condition in question, what immune markers changed in those trials, and whether those marker changes were associated with meaningful clinical improvements in outcomes that matter to patients. Comparing the risk-benefit profile of an ADSC intervention with that of existing approved immunotherapies for the same condition is also essential, and that comparison requires specialist input.
Questions About Tissue Banking and Long-Term Planning
The appropriate framing for tissue banking in the context of adipose immunology is long-term optionality under genuine uncertainty. The science of adipose tissue as an immune organ is robust and well established. The science of ADSC immunomodulation is real and developing. The translation of that science into approved, predictable, scalable immune therapies for specific conditions is a future possibility that requires clinical trial development, regulatory approval, and a manufacturing infrastructure that is still being built. Banking preserves tissue for that future, while acknowledging that the future’s specific form is not yet defined.
Frequently Asked Questions
Why do scientists now call fat an immune organ, not just storage?
Because of the discovery that adipose tissue contains substantial populations of resident immune cells, produces hormones and cytokines that influence immune function throughout the body, and participates actively in immune surveillance and metabolic regulation. A key review accessible at PMC4340740 describes adipose tissue macrophages as central to this dual metabolic-immune role. In lean individuals, adipose immune cells support insulin sensitivity and tissue homeostasis. The fat-as-storage view was accurate but incomplete; the fat-as-immune-organ view adds the biological reality that adipose tissue is in constant immunological conversation with the rest of the body.
What immune cells live in my adipose tissue in a healthy state?
In lean, healthy adipose tissue, the dominant immune cell populations are those with homeostatic and anti-inflammatory functions. These include M2-like macrophages that facilitate lipid turnover and tissue remodeling, eosinophils that contribute to anti-inflammatory signaling through IL-4, innate lymphoid cells type 2 (ILC2s) that maintain anti-inflammatory cytokine production, and regulatory T cells (Tregs) that suppress excessive immune activation. As described in the innate immune cell review at PMC8787575 and the adaptive immune review at Wiley, these populations are not incidental to metabolic function; they actively maintain the insulin-sensitive state of healthy fat tissue.
How does obesity change the immune cell mix inside fat and affect insulin resistance?
In obesity, the expansion and metabolic stress of adipose tissue trigger an influx of pro-inflammatory immune cells. M1-like macrophages accumulate around dying adipocytes, forming crown-like structures, and produce cytokines including TNF-alpha and IL-1 beta that interfere with insulin signaling. Th1 and CD8+ T cells increase while Tregs and Th2 cells decrease, shifting the tissue away from the anti-inflammatory balance of lean tissue. As the review at PMC5199693 describes, these immune changes are mechanistically linked to insulin resistance, not merely correlated with it. The adipose tissue is generating inflammatory signals that reach the liver, muscle, and systemic circulation, propagating metabolic dysfunction beyond the fat depot.
What does it mean when people say adipose-derived stem cells are “anti-inflammatory”?
It means that in experimental systems, including cell culture experiments and animal models, ADSCs secrete molecules that reduce the activity of pro-inflammatory immune cells and promote anti-inflammatory populations like Tregs. The specific mediators include IL-10, TGF-beta, PGE2, IDO, and PD-L1 surface expression. These effects have been documented in peer-reviewed studies, including the comprehensive review at PubMed and the Scientific Reports study at nature.com. The important qualification is that these are experimental findings that do not automatically translate into a claim that ADSCs cure inflammatory diseases in people. Translation requires human trial data, and those data remain preliminary for most conditions.
Can ADSCs reset or cure autoimmune disease by fixing the immune system?
No, not in any established, approved sense. Animal models of autoimmune disease have shown that ADSC administration can reduce inflammation, lower autoantibody production, and improve organ function. Early human trials have explored ADSC safety and immune marker changes in conditions like graft-versus-host disease and other immune-mediated conditions, with generally acceptable safety and some changes in inflammatory markers. But these are early-phase findings that have not produced an approved therapy for any autoimmune disease. The complexity of human autoimmunity, the variability in ADSC biology across donors, and the dose-timing-route challenges in clinical translation all mean that “cure” is not a scientifically supportable description of what these cells can do at this stage.
How do ADSCs interact with macrophages, T cells, and Tregs in studies?
ADSCs promote a shift in macrophages from M1-like (pro-inflammatory) to M2-like (tissue-repair-oriented) activation, primarily through PGE2 and IL-10 secretion. In T cell experiments, they suppress Th1 and Th17 proliferation and promote Treg generation through IDO, TGF-beta, and PD-L1 mechanisms. As the Scientific Reports study at nature.com documents, donor characteristics including sex can influence the strength of these effects. The mechanisms are well described at the in vitro level; whether and to what degree they operate in the complexity of a systemic immune condition in a living patient depends on context, dose, and delivery that are still being characterized in clinical trials.
If I bank my fat, does that change my immune system today?
No. Banking preserves your adipose tissue in cryogenic storage. The banked tissue does not interact with your immune system while frozen, and the banking process itself does not provide any immunomodulatory therapy. Your current immune status is determined by your current biology, your health conditions, your medications, and your lifestyle. Banking is a preservation decision for potential future regulated applications. It is entirely separate from any immune management you are currently undertaking with your healthcare team.
Where can I read more about adipose tissue immunology from reputable scientific sources?
The best primary sources are accessible through PubMed and PubMed Central. The foundational review on adipose tissue as an immunological organ is at PMC4340740. The innate immune cell review is at PMC8787575. The adaptive immune system in fat review is at Wiley. The comprehensive ADSC impact review covering immunomodulation is at PubMed. The patient’s guide to adipose-derived stem cells on this site translates some of this foundational biology into accessible patient language.
Key Takeaways for Patients Thinking About Immunity and Their Fat
Inflammation and the immune system have become central to how many people think about health, and for good reason: they underlie a wide range of chronic conditions. Adipose tissue turns out to be deeply involved in that story, not as a passive bystander but as an active participant in both healthy immune regulation and disease-driving inflammation.
Save My Fat’s goal is to help people understand how adipose tissue and adipose-derived cells interact with the immune system, without turning that science into promises it cannot yet keep.
The grounded picture:
- Adipose tissue is home to innate and adaptive immune cells that support metabolic health in lean states and drive chronic low-grade inflammation in obese ones. This immune biology is real, well established, and clinically relevant.
- Obesity and metabolic stress shift the adipose immune balance toward a pro-inflammatory state that contributes mechanistically to insulin resistance and systemic inflammation, not just as a bystander effect.
- ADSCs can modulate immune responses in experimental settings by affecting cytokines, macrophage polarization, and T cell populations, but that does not make them a general immune cure, and their effects are context dependent, donor variable, and not yet established as effective treatments for specific conditions in adequately powered human trials.
- Banking adipose tissue preserves a cellular resource that has immunomodulatory potential in principle. It does not provide an immune therapy product today, does not replace lifestyle, medications, or specialist-guided care, and does not guarantee access to future immune applications that are still in early development.
When patients encounter claims about “inflammation,” “immune reset,” or “stem cells that fix your immune system,” the framework in this article provides a way to evaluate whether those claims are grounded in the biology or are running ahead of it. The guide to clinical trials for regenerative medicine, the adipose tissue banking guide, and information on pricing, providers, and family banking are available on this site. The about page describes who Save My Fat is.
This article is for educational purposes only and does not constitute medical or legal advice. Legal and medical review including immunology and endocrinology expertise is required before publication. Please consult your healthcare providers for all questions about immune and metabolic health management.
Last Updated: April 28, 2026







