Adipose-Derived Stem Cells in Hair Restoration and Skin Rejuvenation: What Clinical Studies Actually Show
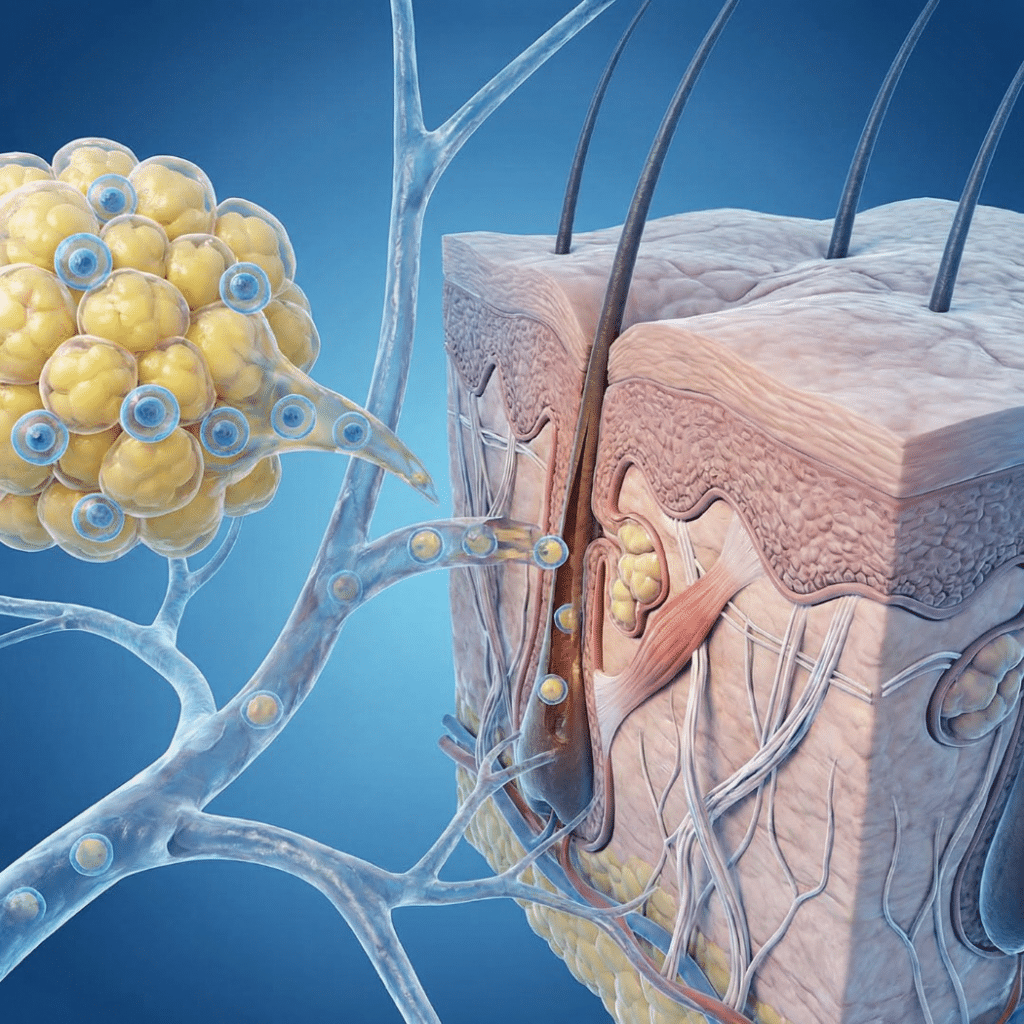
Hair loss affects approximately 50% of men and 40% of women by midlife, and the cosmetic effects of skin aging concern millions more. In recent years, clinics and social media have aggressively promoted “stem cell” and “exosome” treatments for hair regrowth and facial rejuvenation, often with dramatic before-and-after photos and bold promises. The reality is […]
Adipose-Derived Stem Cells in Neurological Conditions: What Clinical Trials Are Investigating
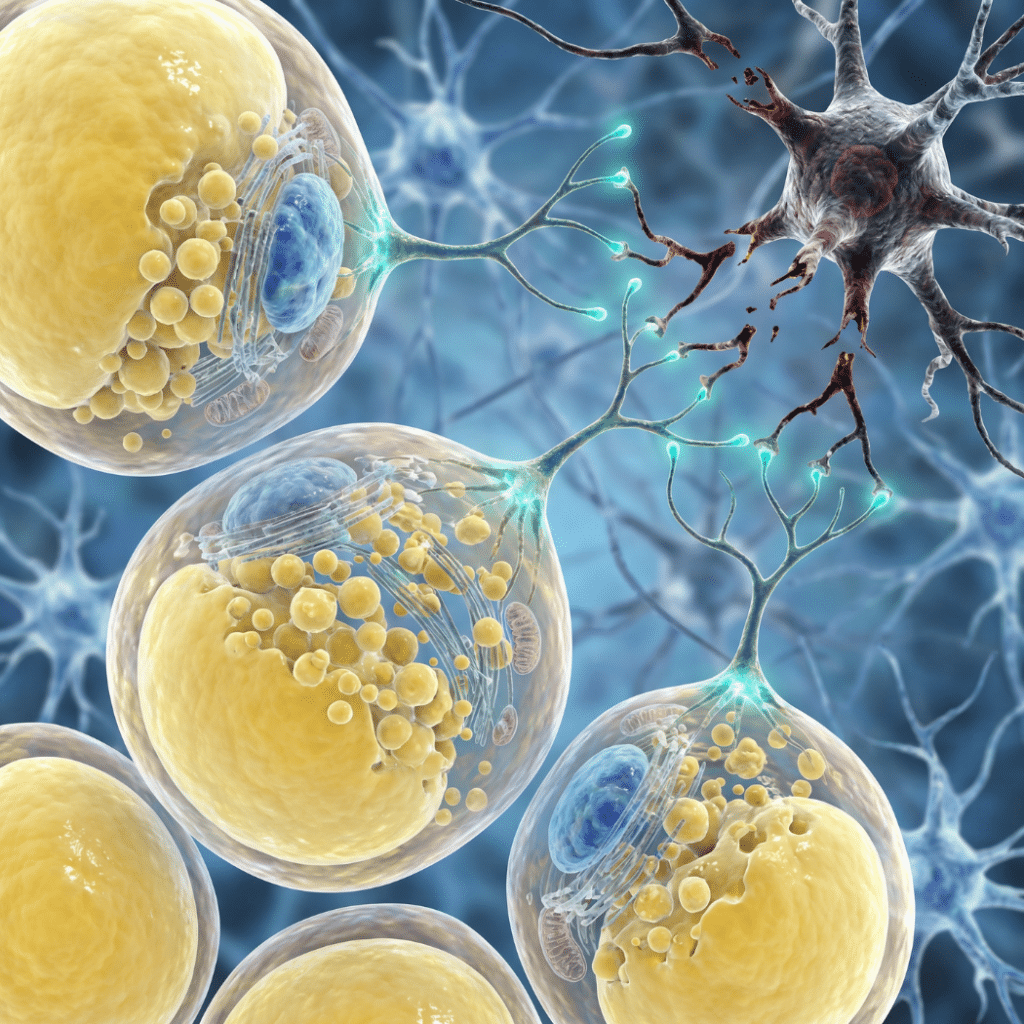
Adipose-Derived Stem Cells in Neurological Conditions: What Clinical Trials Are Investigating Neurological conditions, including spinal cord injury, traumatic brain injury, stroke, Alzheimer’s disease, and multiple sclerosis, represent some of the most challenging areas of modern medicine. The central nervous system has limited capacity for self-repair after injury or disease, and treatment options for many of […]
Adipose-Derived Stem Cells in Wound Healing and Tissue Repair: What Researchers Are Studying

Chronic wounds affect approximately 8.2 million Americans, and diabetic foot ulcers alone are responsible for over 100,000 lower limb amputations each year. Adipose-derived stem cells (ADSCs) and their cell-free derivatives are among the most actively investigated regenerative approaches for wound healing. This guide covers published clinical trial results, 2025 meta-analyses, active trials on ClinicalTrials.gov, emerging […]
Adipose-Derived Stem Cells in Autoimmune and Inflammatory Disease Research: What the Science Shows
Autoimmune and inflammatory diseases affect millions of Americans, and many patients struggle with treatments that provide incomplete relief or carry significant side effects. Researchers are investigating whether adipose-derived mesenchymal stromal cells (ADSCs) could play a role in addressing these complex conditions. This guide walks through the current clinical evidence, the regulatory lessons learned from the […]
Adipose-Derived Stem Cells and Osteoarthritis: What Clinical Trials Are Investigating Right Now
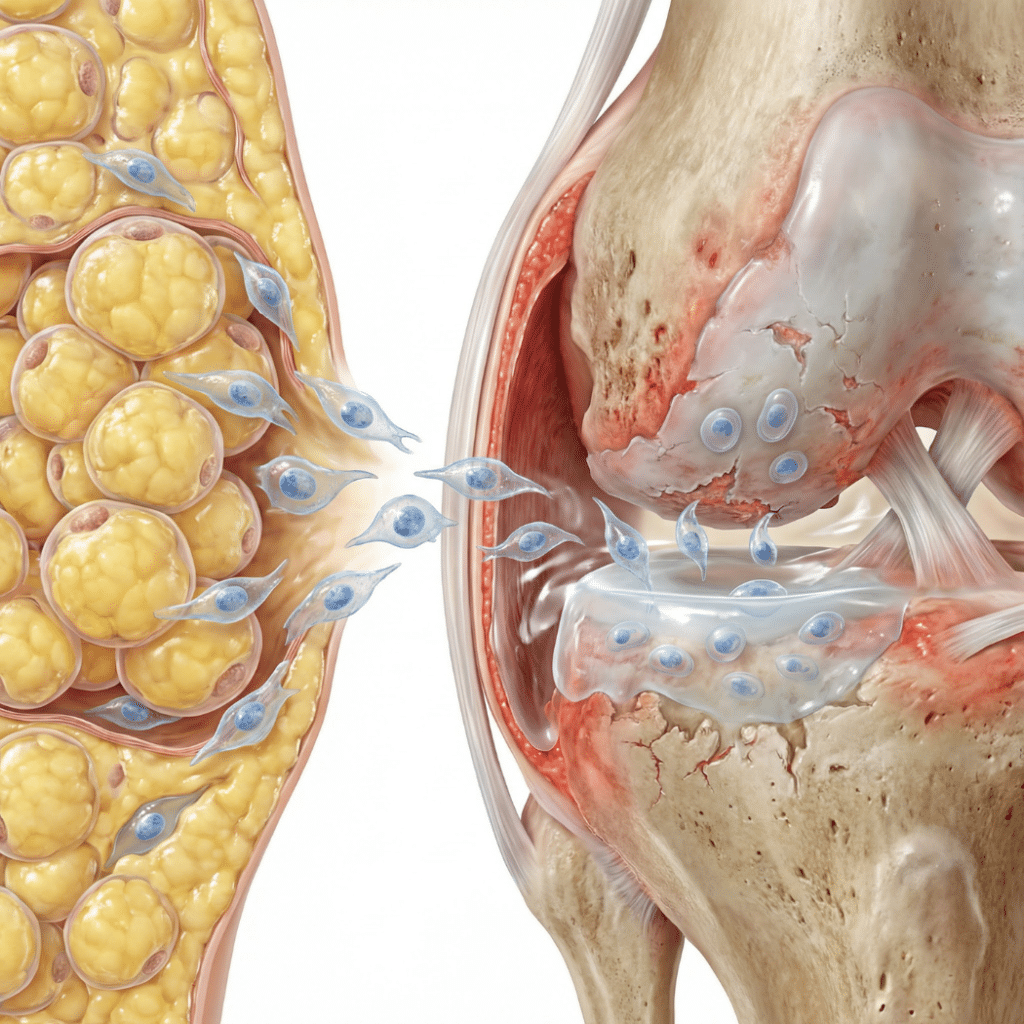
If you are exploring regenerative medicine options for osteoarthritis, understanding the current state of clinical research is essential before making any decisions. This guide provides a comprehensive, science-based overview of clinical trials studying adipose-derived stem cells for osteoarthritis, including trial phases, safety findings, regulatory context, and what this research landscape means for patients considering adipose […]
How to Spot Fake Stem Cell Clinics: Red Flags, FDA Warnings, and How to Find Legitimate Care

Hundreds of clinics across the United States market “stem cell therapy” directly to consumers for conditions ranging from arthritis to Alzheimer’s disease, often charging thousands to tens of thousands of dollars per treatment. Most of these offerings are unproven, unregulated, and have led to documented patient harms, including infections, vision loss, and even deaths. The […]
Family Eligibility for Banked Adipose Tissue: Regulatory Guidelines for First-Degree Relatives

One of the most common questions people ask about adipose tissue banking is whether their family members could potentially use their banked adipose tissue in the future. It is a reasonable question, and the answer involves both biology and regulation. Adipose-derived stem cells (ADSCs) collected from your own tissue are inherently autologous, meaning there is […]
Mesenchymal Stem Cells 101: How Adipose‑Derived Cells Compare with Bone Marrow and Birth Tissue
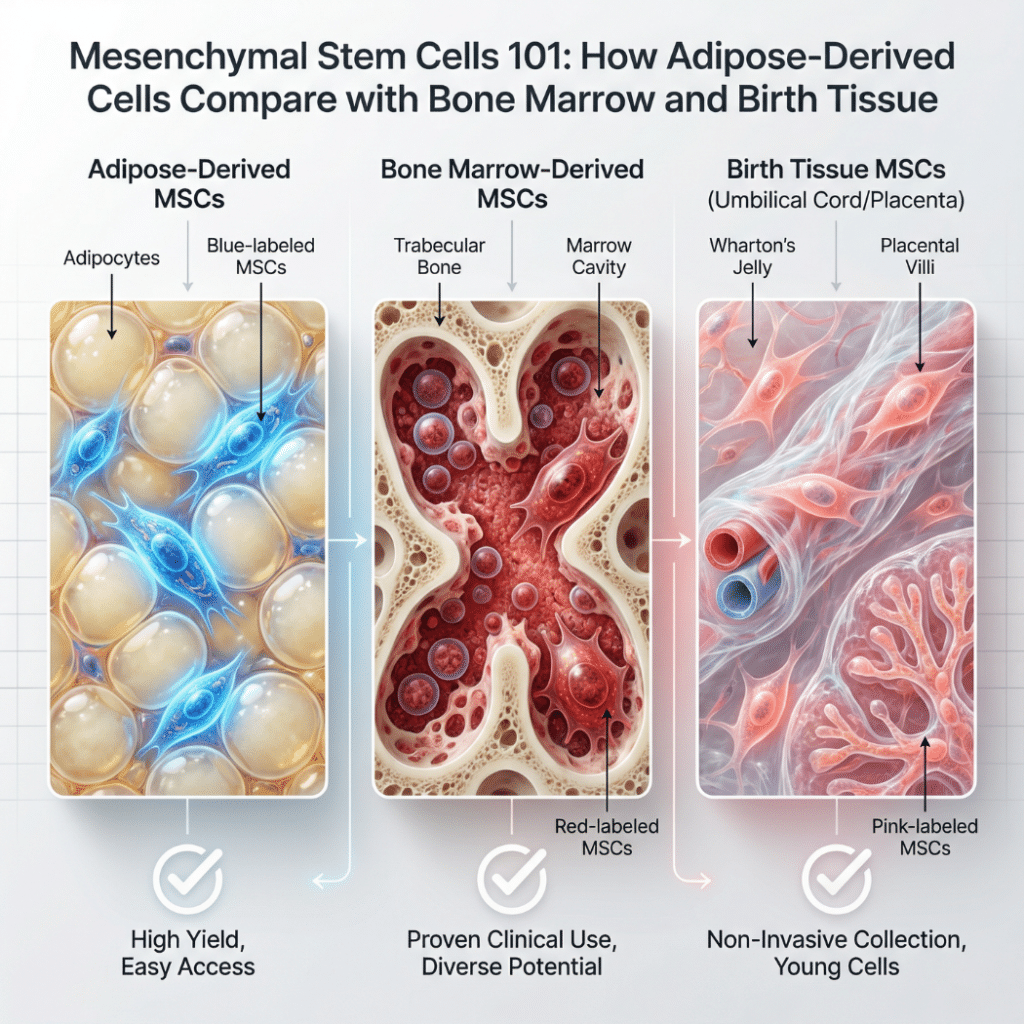
Mesenchymal stem cells, often abbreviated MSCs, are a family of multipotent cells found in bone marrow, adipose (fat) tissue, umbilical cord, and other tissues throughout the body. These cells can differentiate into bone, cartilage, and fat under laboratory conditions and are being studied in hundreds of clinical trials for their regenerative and immunomodulatory properties. Each […]
How to Find Legitimate Clinical Trials In 2026 (Including Stem Cell Studies) Without Getting Scammed

Clinical trials are the primary way new therapies are tested and the only pathway to FDA approval for new drugs and biologics. Many patients with serious or chronic conditions now search for trials on their own, and the volume of information online, especially around stem cell therapies, can make it difficult to distinguish legitimate research […]
Expanded Access Programs: How Patients Can Legally Access Investigational Therapies

Expanded Access, sometimes called compassionate use, is the FDA’s formal pathway for certain patients with serious or immediately life-threatening diseases to access investigational products outside of clinical trials when specific criteria are met. It is a regulated, structured process governed by federal law, and it is distinct from both clinical trials and “Right-to-Try” legislation. This […]

